Eighty-five percent of CNS Tucson cochlear implant recipients say they would do it again — and a new two-minute questionnaire captures the patient experience that traditional speech testing misses.
CHICAGO, May 9, 2026 —
Researchers from Ear & Hearing | Center for Neurosciences (CNS Tucson) presented a study at CI 2026, the 13th annual Conference on Cochlear Implants of the American Cochlear Implant Alliance, held May 6–9, 2026 at the Hyatt Regency Chicago. In Poster #69, “Would You Do It Again? Measuring Global Satisfaction in Cochlear Implant Recipients,” the team introduced and tested a novel 10-item global satisfaction questionnaire — completed by patients in under two minutes — and reported high overall satisfaction across nearly every domain assessed.
Why this study matters: Cochlear implantation is a well-established intervention for adults with severe-to-profound sensorineural hearing loss. Outcomes are traditionally evaluated using objective measures such as speech recognition and aided thresholds. While these metrics are essential, they do not always reflect the patient’s lived experience of overall satisfaction. Existing patient-reported outcome measures (PROMs) typically evaluate discrete domains — communication in noise, listening effort, telephone use, emotional impact — but rarely capture a patient’s global perception of benefit. A simple, clinically meaningful question — Would I choose to undergo cochlear implantation again? — is infrequently assessed. As the field continues to emphasize patient-centered, value-based care, brief tools that assess overall satisfaction are needed. The Center for Neurosciences team designed this study to fill that gap.
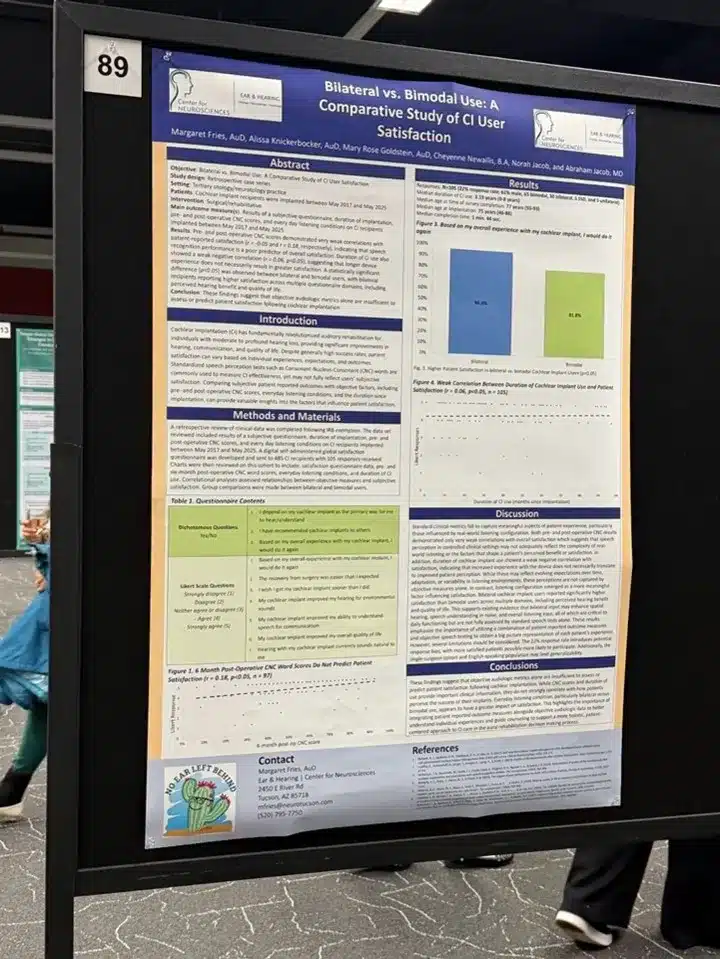
Study design: The investigators developed a 10-item, self-administered global satisfaction questionnaire and distributed it digitally to 485 English-speaking adult cochlear implant recipients implanted between April 7, 2017 and March 30, 2025 by a single surgeon. The survey included seven Likert-scale items (rated from “strongly disagree” to “strongly agree”) and three dichotomous (yes/no) items, including whether patients would choose to receive a cochlear implant again. The survey was designed to take five minutes or less to complete, and participants represented a broad range of ages, device-use durations, speech-recognition abilities, and implant manufacturers. A total of 105 responses (22% response rate; 61% male) were received by the time this study was presented at CI2026. Further data collection is ongoing. Median duration of CI use was 3.19 years (range 0–14 years), median age at implantation was 75 years (range 46–88), and median age at survey completion was 77 years (range 55–93). Median completion time was 1 minute, 44 seconds.
Key findings: When asked the central question — would they undergo cochlear implantation again — 85% of respondents (n = 89) said yes, and 15% (n = 16) said no. On the seven-item Likert scale, 92.4% (n = 97) responded positively or neutrally across the assessed domains, while only 7.6% (n = 8) responded negatively. Seventy-five percent (n = 79) agreed or strongly agreed that their cochlear implant had improved their quality of life. Additional findings underscored the integration of cochlear implants into patients’ daily lives: 76% reported that they depend on their cochlear implant as the primary way they hear and understand, and 75% had recommended cochlear implants to others. Most respondents reported positive experiences across all Likert items, with the strongest gains in communication and quality of life.
Clinical significance for CNS Tucson: The brevity, high completion rate, and strong face validity of the 10-item survey support its feasibility as a practical clinical tool. For the cochlear implant program at Center for Neurosciences — which serves a broad referral base across southern Arizona, including rural communities and a large Veterans Affairs population — a short, digital satisfaction survey can be incorporated into routine post-implantation visits without disrupting clinic workflow. Routine collection of these data has several practical applications: it can support more nuanced patient counseling, set realistic expectations during candidacy discussions, surface dissatisfied patients earlier so that troubleshooting and rehabilitation efforts can be intensified, and provide longitudinal program-level outcomes data that complement standard CNC testing. Together with the team’s companion study on bilateral versus bimodal use (Poster #89, also presented at CI 2026), these data reinforce a central theme: while objective speech-perception testing remains essential, it does not, by itself, capture how patients perceive the success of their implants. Incorporating brief, patient-reported global satisfaction measures into routine cochlear implant care offers a more complete view of outcomes and aligns with the field’s shift toward patient-centered, value-based care.
Study authors: Margaret Fries, AuD; Mary Rose Goldstein, AuD; Alissa Knickerbocker, AuD; Norah E. Jacob; Lexxi-Joy Bruxvoort, BS; and Abraham Jacob, MD. All authors are affiliated with Ear & Hearing | Center for Neurosciences in Tucson, Arizona.
About CI 2026: CI 2026, the 13th annual Conference on Cochlear Implants organized by the American Cochlear Implant Alliance, was held May 6–9, 2026 at the Hyatt Regency Chicago. The multidisciplinary meeting brought together otologists, neurotologists, audiologists, speech-language pathologists, and researchers from across North America and beyond. This year’s program highlighted optimizing clinical efficiency and program growth, mental health comorbidities in CI patients, cognition and aging, care for non-English-speaking populations, and applications of machine learning and artificial intelligence in cochlear implant care.
About Ear & Hearing | Center for Neurosciences. Ear & Hearing is the otology, neurotology, and audiology department at Center for Neurosciences in Tucson, Arizona. The program offers comprehensive evaluation and management of hearing and balance disorders, including a cochlear implant program, skull base surgery, vestibular schwannoma management, stapes surgery, chronic ear surgery, pediatric hearing loss evaluation, ototoxicity monitoring, and amplification with hearing aids. Ear & Hearing serves a broad catchment area across Arizona, New Mexico, Southern Texas, Southern Nevada, parts of Colorado, and even California.
